Amino acids are organic compounds containing an amino group (NH3) and a carboxylic acid group (COOH) that form proteins.
The amino acids within a protein are held together by a covalent peptide bond, forming a long polypeptide chain.
Quick Look
- Amino acids are classified as essential, non-essential, and conditionally essential based on whether the body can naturally produce them.
- There are nine essential amino acids (EAAs) that must be obtained through diet or supplementation.
- All amino acids have the same foundational structure—a carbon backbone bonded to an amine, carboxyl, and side chain.
- Amino acids join via peptide bonds between the carboxyl group of one amino acid and the amino group of another.
What are Amino Acids?
Proteins are one of the most important compounds in the human body. They play a role in everything from hormones and enzymes to growth, repair, and maintenance. But for proteins to be functional, they require the presence of small compounds: amino acids.
In their most basic sense, amino acids are the fundamental building blocks of all proteins. Amino acids are organic compounds composed of three functional groups:
- Amino group (NH3)
- Carboxylic acid group (-COOH)
- R-group (side chain)
When you add an amino group together with a carboxyl (acid) group, you form an amino acid.
While hundreds of amino acids exist in nature, only 20 unique amino acids make up proteins—these are the 20 amino acids that appear in our genetic code.
But these 20 amino acids have different names and functions. So how do you differentiate them?
We’ll cover the structure of amino acids shortly, but the side chain attached to the carbon backbone of an amino acid gives it its specific and unique properties. For example, glycine is the smallest amino acid whose side chain is a single hydrogen atom, while the side chain of leucine is branched and consists of CH2-CH(CH3)2.
3 Types of Amino Acids
Within the 20 amino acids found in the genetic code, they are broken down into three groups:
- Essential amino acids: Amino acids that cannot be produced in the body; these must come from diet or supplementation.
- Non-essential amino acids: Amino acids that can be synthesized in the body from essential amino acids.
- Conditionally essential amino acids: Amino acids that cannot be synthesized in the body in sufficient quantities during specific physiological periods of growth, such as pregnancy, adolescent development, or recovery from trauma. In these situations, they must come from diet or supplements.
Here’s a breakdown of the 20 amino acids in their respective categories.
| Essential | Nonessential | Conditionally Essential |
|---|---|---|
| Phenylalanine | Alanine | Arginine |
| Valine | Aspartic acid (aspartate) | Cysteine |
| Tryptophan | Asparagine | Glutamine |
| Threonine | Glutamic acid (glutamate) | Glycine |
| Isoleucine | Selenocysteine | Proline |
| Methionine | Serine | |
| Histidine | Tyrosine | |
| Leucine | ||
| Lysine |
Key takeaway: Amino acids are the building blocks of all proteins. The body requires the nine essential amino acids to come from diet, but the non-essential and conditionally essential amino acids can be produced in the body.
What Do Amino Acids Do?
The body uses amino acids to produce proteins, but they are also used to synthesize other important nitrogen-containing compounds, including creatine, peptide hormones, and neurotransmitters.
Simply put, amino acids are involved in several essential metabolic functions that enable your body to work correctly.
Here are three benefits of amino acids.
- Support mood
Many nutrients regulate mood, but specific amino acids are especially important. Serotonin, more formally called 5-hydroxytryptamine (5-HT), is a brain chemical that regulates various aspects of normal function, including mood, appetite, sleep, and other behaviors.
The body requires tryptophan to produce serotonin:
Tryptophan — 5-Hydroxytryophan — 5-Hydroxytryptamine, 5-HT (serotonin)
Low serotonin levels have been linked to several health concerns, including depression and disrupted sleep. However, research shows that supplementing with tryptophan before bed can reduce symptoms of depression and anxiety and improve sleep quality.
- Enhance athletic performance and recovery

Want to build bigger muscles? Looking for a way to accelerate muscle recovery post-workout? Try essential amino acids! Because amino acids are the building blocks of protein, your body requires them to build and maintain muscle and support a more efficient recovery.
Specifically, the branched-chain amino acids (BCAAs)—leucine, isoleucine, and valine—play integral roles in stimulating muscle protein synthesis. A study published in Frontiers of Physiology found that people who consumed 5.6 grams of BCAAs post-workout saw 22% more muscle protein synthesis compared to people taking a placebo.
But it’s not just muscle growth and maintenance that amino acids are involved in—they also play an important role in muscle recovery. Studies have shown that BCAA consumption can attenuate muscle damage by reducing muscle protein breakdown during exercise and decreasing levels of creatine kinase, an indicator of muscle damage. So, taking BCAAs or EAAs pre- or post-workout may alleviate delayed onset muscle soreness (DOMS) and get you back to training faster.
- May promote better sleep quality

Are you sick and tired of restless, non-rejuvenating sleep? Add some amino acids to your diet. There are three main amino acids that are sleep-supportive:
- Glycine: Glycine has been shown to support sleep by supporting a reduction in core body temperature and helping you reach a deeper sleep stage faster.
- GABA: Activation of GABA receptors in the brain favors sleep by reducing brain activity and making it easier to fall and stay asleep.
- Tryptophan: The precursor to melatonin production. Supplementing with this amino acid may improve sleep onset latency.
Key takeaway: Essential amino acids play important roles in supporting mood through serotonin production, enhancing exercise performance and recovery, and promoting better sleep quality.
Amino Acid Structure
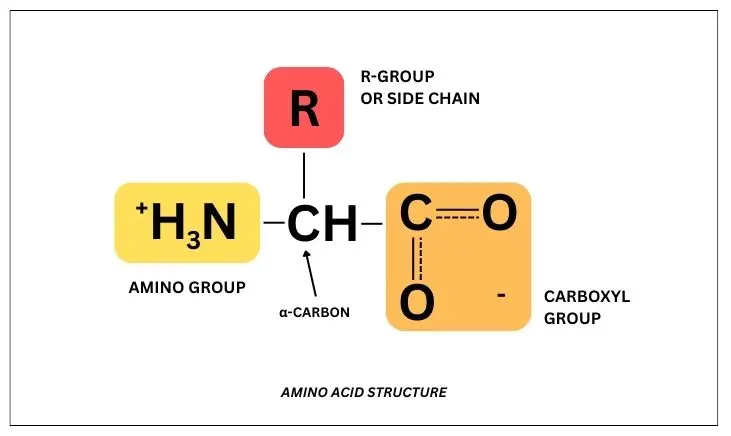
All 20 amino acids have the same foundational structure—they have a carbon backbone attached to three functional groups:
- Amino (NH3)
- Carboxyl (COOH)
- R-group (side chain)
But what actually differentiates amino acids? Their side chains or R-groups.
Let’s look at an example.

The simplest amino acid is glycine—its side chain is a single hydrogen atom. Compare that to serine, whose side chain consists of a carbon bonded to an alcohol (CH2OH).
That said, side chains aren’t the one thing that differentiates one amino acid from another. They’re also categorized based on individual properties, including:
- Charge (positive or negative)
- Hydrophobicity (attract or repel water)
- Polarity (polar or non-polar)
These properties of each amino acid influence how amino acids interact with other amino acids and their three-dimensional structure and properties.
Key takeaway: All amino acids contain an amino group (NH2), a carboxyl group (COOH), and a unique side chain specific to that amino acid.
How Do Amino Acids Bond Together?
Amino acids contain functional groups that differentiate them from one another, but what binds amino acids together in a protein?
Covalent bonds called peptide bonds.
Peptide bonds are essential links that connect amino acids to form polypeptide chains that eventually fold into a specific conformation, depending on how amino acids interact with one another, to form a functional protein.
A peptide bond forms when the carbon atom from the carboxyl group of one amino acid shares electrons with the nitrogen atom from the amino group of another amino acid. In this condensation reaction, a water molecule is released.

The repeating amino acid sequence forms the polypeptide backbone of the protein or molecule. Attached to this chain are the parts of the amino acids that aren’t involved in making peptide bonds, which give the amino acid its unique properties—the 20 different amino acid R-groups or side chains. As mentioned, these R-groups have specific properties; they can be polar/non-polar, hydrophilic or hydrophobic, positively or negatively charged, and so on.
Bonded amino acids joined by peptide bonds are named for the number of amino acids bonded together:
- Two amino acids = dipeptide
- Three amino acids = tripeptide
- Four amino acids = tetrapeptide
- Five amino acids = pentapeptide
And so on…
Chains with multiple amino acids ranging from two to twenty are typically classified as oligopeptides. In comparison, more than twenty amino acids are generally classified as polypeptides, although this term is used for chains of unspecified length. Fifty or more amino acids linked together by peptide bonds are typically classified as proteins.
Order of Protein Structure
Before we wrap up, there’s one other area to touch on with proteins and peptide bonds—the order of protein structure.
The shape of a protein is essential to its function, but to understand how a protein gets its shape, it’s important to first understand the four levels of protein structures: primary, secondary, tertiary, and quaternary.
Primary Structure
The primary structure of a protein is its sequence of amino acids linked together to form a polypeptide chain or protein molecule. During the protein biosynthesis process, each individual amino acid binds to another amino acid via a peptide bone. The two ends of the polypeptide chain are called the amino terminus (N-terminus) and the carboxyl terminus (C-terminus).
Secondary Structure

The secondary structure of a protein refers to the hydrogen bonds formed between atoms of the polypeptide backbone that create local folded structures. The most common types of secondary structures include the α-helix and the β-pleated sheet. Hydrogen bonds hold both of these structures in shape.
Hydrogen bonds form between two atoms:
- Partially negative oxygen atom of the carboxyl group
- Partially positive hydrogen atom of the amino group
Note: Hydrogen bonds involved in the secondary structure of a protein do not include any amino acid side chains (R-groups).
Tertiary Structure

The tertiary structure of a protein refers to the overall three-dimensional shape formed when the side chains of various amino acids interact. These R-groups have varying properties—polar, non-polar, charged, hydrophobic, etc.—which influence how they interact with other amino acids and the resulting 3D structure or overall shape of the protein.
We are not going to get into the specifics of the tertiary structure of a protein, but it’s important to know that several interactions stabilize it, including:
- Disulfide linkages
- Salt bridges
- Coordinate bonds with metal ions
- Hydrogen bonding
- Hydrophobic interactions
Quaternary Structure

Lastly, we have the quaternary structure of a protein, which refers to the association or interaction of several protein chains or subunits into a closely packed arrangement. Each subunit has its own primary, secondary, and tertiary structure, and the subunits are bonded together via hydrogen bonds and van der Waals forces between nonpolar side chains.
For example, hemoglobin, the oxygen-carrying molecule in the bloodstream, is one example of a protein with a quaternary structure. It is made up of four subunits, two of the α and two of the β types.
Note: Not every protein has a quaternary structure—only proteins that are made up of multiple polypeptide chains (subunits) will come together to produce a quaternary structure.
Conclusion
Amino acids are organic compounds that form the foundation of functional proteins. They play important roles in regulating mood, muscle growth and recovery, and sleep, but they also form hormones and enzymes, improve digestion and gut health, and more. Every amino acid contains a carboxyl group (COOH), an amino group (NH2), and an R-group (side chain that’s unique to each amino acid). When amino acids link, the amino group of one amino acid links to the carboxyl group of another amino acid via a peptide bond to form a polypeptide.
FAQs
How do amino acids bond together?
Peptide bonds join amino acids together. They form between the amino group of one amino acid and the carboxyl group of another, releasing a water molecule in the process. The peptide bond formation is, therefore, classified as a dehydration reaction.
How are amino acids connected to form a chain?
Amino acids are connected through a dehydration reaction between two amino acids. The alcohol (carboxyl group) of one amino acid joins with the hydrogen atom (amino group) of another amino acid to release a water molecule and form a peptide bond.
Do free amino acids have peptide bonds?
No, free amino acids do not have peptide bonds. Free amino acids function as monomers and do not have any peptide linkages to other amino acids. In the case of proteins, linked amino acids do contain peptide bonds to connect them.
When amino acids bond together, what is formed?
When amino acids bond together via peptide bonds, they form polypeptides. When 50 or more amino acids bond together, they are classified as proteins. In the process of the dehydration reaction, a water molecule is also formed.
What is the difference between tertiary and quaternary proteins?
The tertiary structure of proteins refers to the overall 3D structure of the protein, whereas the quaternary structure is the structure that arises when separate chains of proteins aggregate with self or other proteins.