Amino acids contain an acid group–a carboxylic acid–which would suggest all amino acids are acidic, but that’s not always the case. Amino acids can be acidic or basic depending on the pH of their side chains. Acidic amino acids have acidic side chains, while basic amino acids have basic side chains. Aspartate (aspartic acid) and glutamate (glutamic acid) are acidic amino acids, while arginine, histidine, and lysine are basic amino acids.
Quick Look
- Amino acids are organic compounds with an amino group (NH2), a carboxyl group (COOH), and an R-group (side chain) that varies by amino acid.
- There are nine essential amino acids (EAAs) that cannot be synthesized in the body and 11 non-essential amino acids that can be synthesized from EAAs.
- Most amino acids exist as zwitterions—ions that contain two functional groups and possess positive and negative electrical charges, making them electrically neutral.
- Acidic amino acids contain acidic side chains, such as aspartate (aspartic acid) and glutamate (glutamic acid).
- Basic amino acids contain basic side chains, such as arginine, histidine, and lysine.
The Basics of Amino Acids
Twenty essential and non-essential amino acids form the foundation of all functional proteins in the human body. Amino acids are organic compounds that contain an amino (NH2) and carboxylic acid (COOH) functional group.
Although most amino acids are involved in protein synthesis to some degree–these are called proteinogenic amino acids–they also serve several other roles. Some of the functions of amino acids include:
- Synthesis of nitrogen-containing compounds, including creatine, peptide hormones, and neurotransmitters
- Hormone and enzyme synthesis
- Tissue growth and repair
- Energy production
- Immune function
- Nutrient absorption
While hundreds of different amino acids exist in nature, only 20 are found in our genetic code—these are the essential and non-essential/conditionally essential amino acids.
Amino Acids: Essential vs. Nonessential
Amino acids are categorized based on whether they can be synthesized endogenously or must come from external sources.
- Essential amino acids: Amino acids that cannot be synthesized endogenously and are dietarily essential.
- Non-essential amino acids: Amino acids that can be synthesized endogenously from essential amino acids.
- Conditionally essential amino acids: Amino acids that cannot be synthesized in the body in sufficient quantities during specific physiological periods of growth, such as pregnancy, adolescent development, or recovery from trauma. In these situations, they must be obtained through diet or essential amino acid supplements.
Here’s a breakdown of the 20 amino acids in their respective categories.
| Essential | Non-essential | Conditionally essential |
|---|---|---|
| Phenylalanine | Alanine | Arginine |
| Valine | Aspartic acid (aspartate) | Cysteine |
| Tryptophan | Asparagine | Glutamine |
| Threonine | Glutamic acid (glutamate) | Glycine |
| Isoleucine | Selenocysteine | Proline |
| Methionine | Serine | |
| Histidine | Tyrosine | |
| Leucine | ||
| Lysine |
Key takeaway: Amino acids form the foundation of all functional proteins. They are categorized as essential, non-essential, and conditionally essential based on whether they can be synthesized in the body.
Amino Acid Structure
The properties and functions of amino acids vary based on their structure. While the “core” of every amino acid is the same, there is a fundamental difference in structure that differentiates one from another.
All amino acids are composed of four constituents:
- Central carbon (alpha or chiral carbon)
- Amino group (-NH2)
- Carboxyl group (-COOH)
- R-group (side chain)
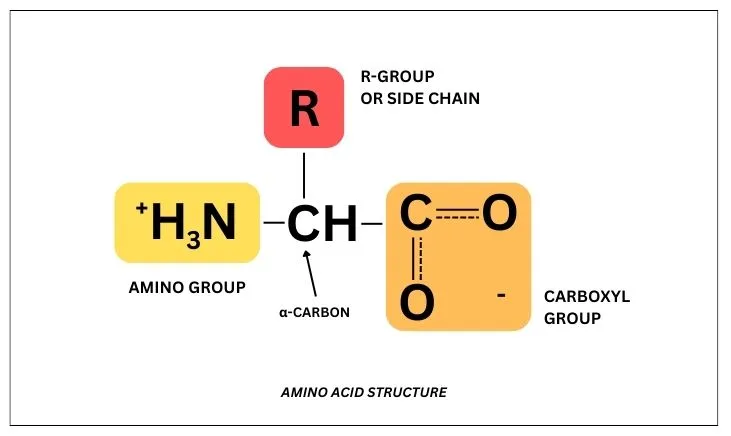
While the alpha carbon, amino group, and carboxyl group remain constant between all 20 amino acids, the R-group is what changes.
For example, the R-group in glycine, the smallest and simplest amino acid, is a single hydrogen atom. In contrast, the R-group in tryptophan, a much larger amino acid, is an indole–an aromatic, heterocyclic, organic compound with the formula C8H7N.
Take a look at their structural differences:

The R-group isn’t the only factor that differentiates amino acids. They are also categorized by the following:
- Charge (positive or negative)
- Hydrophobicity (attract or repel water)
- Polarity (polar or non-polar amino acids)
These properties influence how amino acids interact with other amino acids in polypeptides and proteins, ultimately affecting their 3D structure and properties.
Acidic vs Basic Amino Acids

The terms acid and base refer to the pH of a specific compound. The pH scale ranges from 0 to 14 and is as follows:
- pH 7 = neutral
- pH <7 = acid
- pH >7 = base
So, what does it mean to be an acidic or basic amino acid?
The structure of an amino acid allows it to act as an acid or a base, depending on what it’s reacting with. This is because at a certain pH value–this varies based on the amino acid–most amino acids exist as zwitterions; zwitterions are an ion that contains two functional groups. Zwitterions are ions that possess both positive and negative electrical charges, making them electrically neutral. If an acid is added to a solution containing the zwitterion, the carboxyl group captures a hydrogen (H+) ion, and the amino acid becomes positively charged. If a base is added, the H+ ion is removed from the amino group of the zwitterion, producing a negatively charged amino acid.
Regardless of whether an acid or base is added, the amino acid maintains the pH of the system—that is, it removes the added acid (H+) or base (OH−).
Here’s an example:

The pH at which an amino acid exists in solution as a zwitterion is called the isoelectric point (pI). At its pI, the positive and negative charges on the specific amino acid balance, and the molecule becomes electrically neutral.
Amino acids with neutral side chains have isoelectric points ranging from 5.0 to 6.5. Basic amino acids, which have positively charged R-groups at a neutral pH, have relatively high examples. On the other hand, acidic amino acids, which have negatively charged R-groups at a neutral pH, have low examples.
The table below summarizes examples of some amino acids:
| Amino acid | Classification (polar, nonpolar, charged) | pI |
|---|---|---|
| Alanine | Nonpolar | 6.0 |
| Arginine | Positively charged (basic) | 10.8 |
| Aspartic acid | Negatively charged (acidic) | 3.0 |
| Glutamic acid | Negatively charged (acidic) | 3.2 |
| Histidine | Positively charged (basic) | 7.6 |
| Lysine | Positively charged (basic) | 9.8 |
| Serine | Polar, uncharged | 5.7 |
| Threonine | Polar, uncharged | 6.5 |
| Valine | Nonpolar | 6.0 |
Key takeaway: Amino acids can act as acids or bases due to the presence of the amino and carboxyl functional groups. The pH at which an amino acid exists in solution as a zwitterion is called the isoelectric point (pI).
What Are Acidic Amino Acids?
Understanding acids and bases is a chemistry lesson, but we’re trying to keep it simple.
So, of the essential and non-essential amino acids, what are basic amino acids, and what are acidic amino acids?
The acidity of amino acids is measured with what’s called a pKa number. It’s obtained using a logarithm and measures the acid strength, while pH measures the hydrogen content of a solution. Acids with a smaller pKa number are more acidic and, therefore, have a lower pH value.
Acidic Amino Acids
There are two acidic amino acids:
- Aspartate (aspartic acid)
- Glutamate (glutamic acid)
Acidic amino acids are those with acidic side chains, specifically those containing carboxylic acid functional groups with pKa values low enough to lose protons and become negatively charged. By nature, acidic amino acids are also hydrophilic, meaning they like water, and polar, meaning they are positively charged.
Basic Amino Acids
There are three basic amino acids:
- Arginine
- Histidine
- Lysine
These amino acids are basic because they have nitrogen-containing side chains resembling ammonia, a base. The pKa values of basic amino acids are high enough to bind protons, making them positively charged. By nature, basic amino acids are polar and hydrophilic.
Conclusion
Understanding acids and bases in amino acids can be tricky, but here’s something to remember: the difference between acidic and basic amino acids is no different than any acid or base in chemistry. All amino acids have the same basic structure and are electrically neutral, but their side chains can be acidic or basic, making the amino acid acidic or basic.



